| CPC A61K 38/29 (2013.01) [A61K 9/00 (2013.01); A61K 47/34 (2013.01); A61K 47/60 (2017.08)] | 12 Claims |
|
1. A method of treating a human patient having hypoparathyroidism comprising subcutaneously administering to the subject a PTH compound, wherein the pharmacokinetic profile of the PTH compound exhibits a peak to trough ratio of less than 4 within one administration interval at a steady state,
wherein the PTH compound is a water-soluble controlled-release PTH compound of formula (Ia) or a pharmaceutically acceptable salt thereof:
 wherein
-D is a PTH moiety, which has the sequence of SEQ ID NO: 51;
-L2-L1- has the formula:
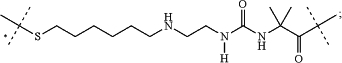 wherein the unmarked dashed line indicates the attachment to a nitrogen of -D by an amide bond; and the dashed line marked with an asterisk indicates attachment to —Z;
x is 1;
—Z comprises a moiety of formula (b):
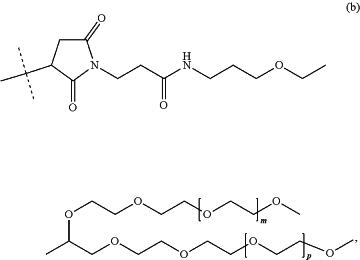 wherein the dashed line indicates attachment to -L2 and m and p are independently of each other an integer ranging from 150-1000; and
the PTH moiety is released with a release half-life of at least 12 hours.
|